|
11/10/2023 0 Comments Periodic table zn elementAdditional Notes: Many sources of European origins credit Andreas Marggraf with discovering zink in 1746, however, it had been know in India and China since before the 1500s.Used in alloys such as brass, bronze, nickel. Uses of Zinc: Used to coat other metals (galvanizing) to protect them from rusting. 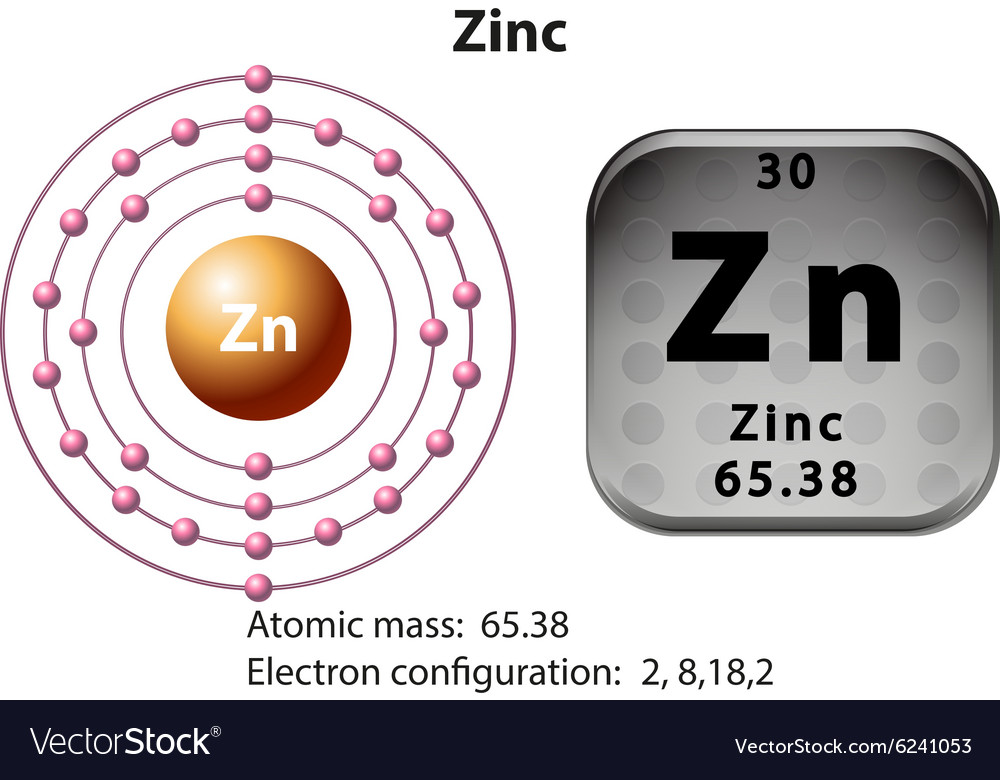
Primary mining areas are USA, Canada, Australia, Austria, Russia and Turkey. Annual world wide production is around 5,020,000 tons. Sources of Zinc: Found in the minerals zinc blende (sphalerite) (ZnS), calamine, franklinite, smithsonite (ZnCO 3), willemite, and zincite (ZnO).Name Origin: German: zink (German for tin).Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Vapor Pressure = C Regulatory / Health.Enthalpy of Vaporization: 115.5 kJ/mole.Enthalpy of Atomization: 129.7 kJ/mole 25☌.Description: Hard, brittle, shiny bluish-white transition metal.Conductivity Electrical: 0.166 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 25E -6.Valence Electron Potential (-eV): 38.9 Physical Properties of Zinc.Electronegativity: 1.65 (Pauling) 1.66 (Allrod Rochow).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
